Unit Circle
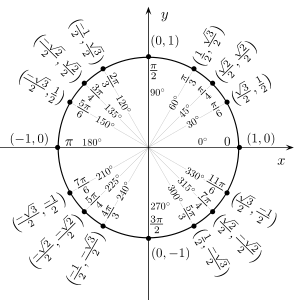
Complex numbers can be identified with points in the Euclidean plane, namely the number a + bi is identified with the point (a, b). Under this identification, the unit circle is a group under multiplication, called the circle group. This group has important applications in mathematics and science. See here.
Just briefly showing containment of “collision process” and for later study. This is how I see this “relation of cosmic particle collisions” to incidents in “high energy collisions processes” and I wonder if this is wrong? Also pointing toward “Neutrino oscillation” as a probabilistic consequence of Quantum mechanics.
To the Substance of this Post
Q9 raised an issue that is of some significance to me because of the way I was “geometrically seeing these collision processes.” While, I had not moved my thinking to the human factor in this process, it has in my study raised the question of what effect it has on the human populations on a personal note.
Quasar9:
The “>general effects of radon to the human body are due to its radioactivity and consequent risk of radiation-induced cancer. As an inert gas, “radon has a low solubility in body fluids which lead to a uniform distribution of the gas throughout the body” (Lindgren, 1989). Radon gas and its solid decay products are carcinogens. Some of the daughter products, especially polonium-218 and 214, from radioactive decay of radon present a radiologic hazard. Depending on the size of the particles, radon decay products can be inhaled into the lung where they undergo further radioactive decay releasing small bursts of energy in the form of alpha particles that can either cause double strand DNA breaks or create free radicals that can also damage the DNA. Also See: Radon
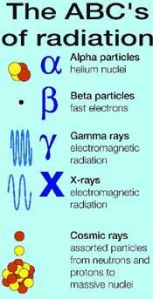
The ABCs – and Xs and Zs – of Radiation
Alpha and beta rays are particles. Gamma rays are electromagnetic radiation, like X-rays but at higher energies. Health physicists worry most about HZE cosmic rays, those with high mass (Z stands for atomic number, which also implies mass) and energy (E). They have two principal sources, the Sun and the galaxy.
Quasar9:
The energy of alpha particles varies, with higher energy alpha particles being emitted from larger nuclei, but most alpha particles have energies of between 3 and 7 MeV. This is a substantial amount of energy for a single particle, but their high mass means alpha particles have a lower speed (with a typical kinetic energy of 5 MeV the speed is 15,000 km/s) than any other common type of radiation (β particles, γ-rays, neutrons etc). Because of their charge and large mass, alpha particles are easily absorbed by materials and can travel only a few centimetres in air. They can be absorbed by tissue paper or the outer layers of human skin (about 40 micrometres, equivalent to a few cells deep) and so are not generally dangerous to life unless the source is ingested or inhaled. Because of this high mass and strong absorption, however, if alpha radiation does enter the body (most often because radioactive material has been inhaled or ingested), it is the most destructive form of ionizing radiation. It is the most strongly ionizing, and with large enough doses can cause any or all of the symptoms of radiation poisoning. It is estimated that chromosome damage from alpha particles is about 100 times greater than that caused by an equivalent amount of other radiation. The alpha emitter polonium-210 is suspected of playing a role in lung and bladder cancer related to tobacco smoking. Also See:Alpha Particles
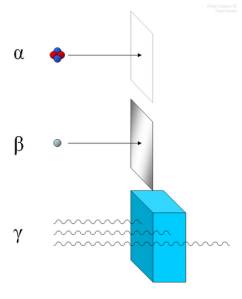
Low energy alpha particles may be completely stopped by a sheet of paper, beta particles by aluminum shielding. Gamma rays, being very high energy in nature, can only be reduced by much more substantial obstacles, such as a very thick piece of lead.
As for types of radioactive radiation, it was found that an electric or magnetic field could split such emissions into three types of beams. For lack of better terms, the rays were given the alphabetic names alpha, beta, and gamma, names they still hold today. It was immediately obvious from the direction of electromagnetic forces that alpha rays carried a positive charge, beta rays carried a negative charge, and gamma rays were neutral. From the magnitude of deflection, it was also clear that alpha particles were much more massive than beta particles. Passing alpha rays through a thin glass membrane and trapping them in a discharge tube allowed researchers to study the emission spectrum of the resulting gas, and ultimately prove that alpha particles are in fact helium nuclei. Other experiments showed the similarity between beta radiation and cathode rays; they are both streams of electrons, and between gamma radiation and X-rays, which are both high energy electromagnetic radiation.
Although alpha, beta, and gamma are most common, other types of decay were eventually discovered. Shortly after discovery of the neutron in 1932, it was discovered by Enrico Fermi that certain rare decay reactions give rise to neutrons as a decay particle. Isolated proton emission was also eventually observed in some elements. Shortly after the discovery of the positron in cosmic ray products, it was realized that the same process that operates in classical beta decay can also produce positrons (positron emission), analogously to negative electrons. Each of the two types of beta decay acts to move a nucleus toward a ratio of neutrons and protons which has the least energy for the combination. Finally, in a phenomenon called cluster decay, specific combinations of neutrons and protons other than alpha particles were found to occasionally spontaneously be emitted from atoms.